First described in the literature in 1908! For egg. Noon and Freeman described grass immunotherapy in 1911. First double-blind trial was by William Frankland in 1954 for subcutaneous grass immunotherapy for seasonal asthma.
Should always be done with extreme caution, if at all, if asthma. Available for wasp/bee stings, grass and tree pollen. Lots of evidence for food allergies, especially in younger children eg peanut immunotherapy. House dust mite sublingual now approved by NICE for resistant allergic rhinitis.
Some evidence that immunotherapy for rhinitis also prevents asthma, which fits with “one airway” hypothesis.
Immunotherapy should be initiated and monitored in a specialist centre experienced in immunotherapy.
Text message reminders doubles adherence – even non-personalised.
Immunomodulation with omalizumab appears to improve success rates of immunotherapy. Treatment with dupilumab (anti-IL4/13) reduces specific IgE levels in people with atopic dermatitis (clinical effect unknown just now).
Grass/pollen allergy
EAACI indications for rhinoconjunctivitis immunotherapy includes:
- mild rhinitis for reasons of asthma control.
- Moderate-severe symptoms.
Bad asthma and poor compliance are contraindications. Bad asthma was an exclusion criterium for most of the studies, so no evidence of safety. Poor evidence for under 5yrs (for HDM).
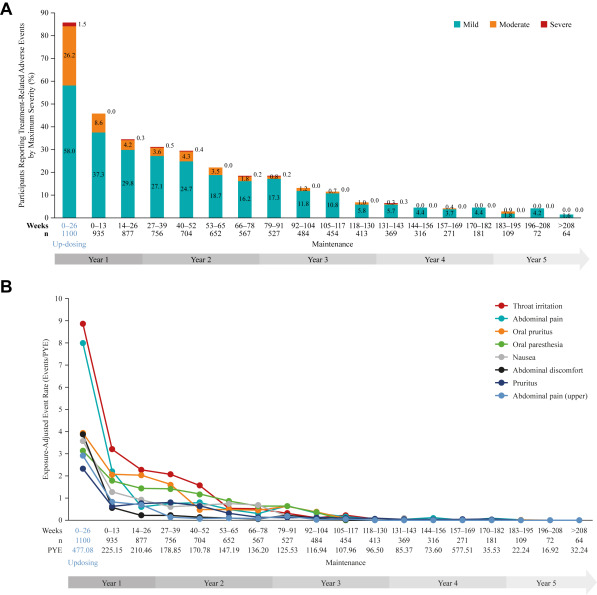
Polysensitization common, US tend to to treat all, Europe tends to pick 1 or 2 most useful, at intervals. 17 fatalities to date with immunotherapy. 1 per million injections (risk with sublingual much less, even though used for higher risk patients). Large local reactions common but don’t predict further reactions. Risk of systemic reaction only increased if recurrent. Consider predosing with antihistamine, else reduce dose.
Sublingual immunotherapy (SLIT) in children aged 4 to 12 years with grass pollen-allergic rhinitis/rhinoconjunctivitis significantly reduced symptoms and medication use, well tolerated, and no serious treatment-related events were reported. [Journal of Allergy & Clinical Immunology. 130(4):886-93.e5, 2012 Oct.]
In metanalysis by Dhami S et al, overall standardized mean difference (SMD) of -0.53 (95% CI -0.63, -0.42) in symptoms scores, roughly equal numbers of SCIT and SLIT studies, roughly equivalent scores. When looking just at children, benefit seems less (SMD -0.25 (95% CI -0.46, -0.05)). Continuous treatment probably slightly better than pre/co-season treatment. Manufacturers suggest “disease modifying effect” of treatment beyond first year, which has theoretical rationale. Four studies of long term outcome, demonstrates continuing benefit if treatment continued beyond first year [Allergy 2017].
Grazax (grass) can be safely administered by general practitioners (£80 per month, licensed from 5yrs up): tablet needs to be kept under tongue for at least 1 minute, first dose should be monitored by doctor for 20 minutes. Don’t eat or drink for 5 minutes.
Give antihistamines for local effects. Oral blistering occurs! Isolated cases of eosinophilic oesophagitis but impossible to link of course.
Contains fish gelatine but no reported problems in those with fish allergy. Severe asthma contraindication (in children, defined as <80% predicted FEV1 on treatment).
Aim to start 4 months before season starts, although still some benefit if started 2-3 months before. If no benefit in first season, no point continuing (according to Grazax own SPC).
Symptom relief begins only in the second season of therapy? BNFc says continue only for 3 yrs. Not approved by SMC for Scotland, because no great evidence for benefit after first year – would need individual patient treatment request. Rosie says children wouldn’t tolerate daily doses for months and years.
Pollinex subcut, given into middle third of upperarm. 2 versions: Trees, and 13 grasses incl rye. 3 injections at 7-14 day intervals each year (£450 each), before season starts. Maintenance kit of 4 vials also available, presumably if additional benefit thought possible. Manufacturer recommends using for 3 successive years. Asthma and beta blocker treatment are relative contraindications. Age 6+, not in SMC at all. [Metanalysis, Chest 2008; 133:589, Journal of Allergy & Clinical Immunology. 115(4):676-84, 2005 Apr.]
Some evidence for short course – in multicentre study of adults (not UK) with grass mix SPT positive rhinitis, 8 subcutaneous injections of placebo or Lolium perenne (LPP) were administered in 4 visits (2 jabs each visit, 30 mins apart, different arms) over 3 weeks between January and April. Combined symptom and medication score (CSMS) measured over the peak pollen season was reduced 15.5% (P = .041) during the peak period and −17.9% (P = .029) over the entire pollen season. Also lower rhinoconjunctivitis QOL global score (P = .005) compared with placebo group.
Asthma
Dhami metanalysis immunotherapy for asthma in kids – symptoms improved, less medication, esp HDM, grass, cat/dog. Not just severe asthma! But no prolonged benefit.
1 study of adults with poor control, HDM. Reduces time to first exacerbation.
Mite allergy prevention study n=111 HDM treatment effective. 2002 PAT study reduced asthma at 10yr follow up after grass/pollen treatment.
[Gillian Vance, Newcastle]
Eczema
House dust mite immunotherapy with SLIT (3 doses per week) shown to have some benefit in RCT from Brazil (66 children and adults) using SCORAD eczema severity questionnaire. Placebo group showed 35% improvement over 18 months, SLIT group showed 55%. No difference in Dermatology Life Quality index, pruritus score or any of the various other measures used!