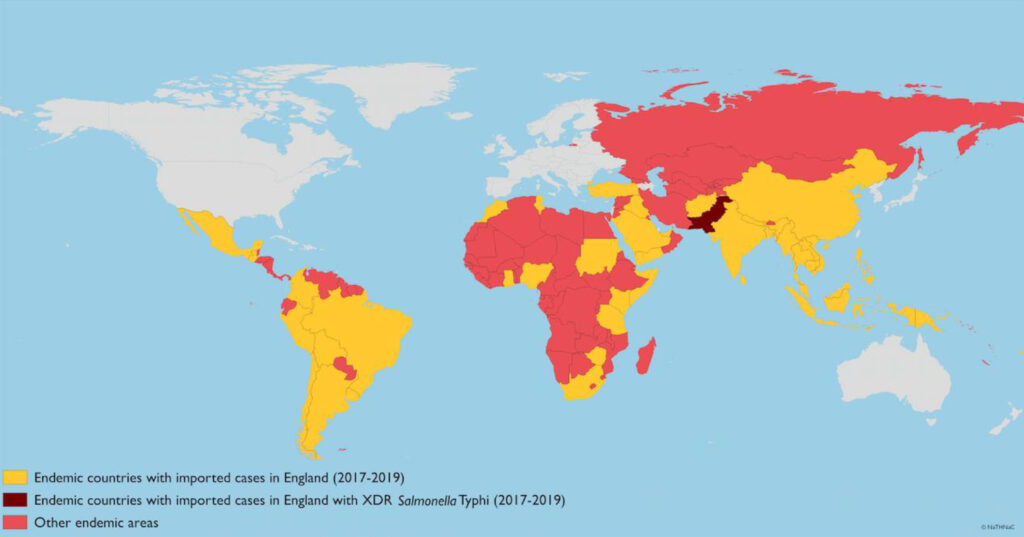
=inflammation of the meninges. Clinically fever, neck stiffness, headache, altered consciousness. Photophobia is classic, but not one of the NICE red flags. Almost always vomiting. Fever can be absent, particularly in young babies, or masked by antipyretics. Can be viral, bacterial or tuberculosis.
2024 NICE guidance (adults and children).
This clinical picture gets confused with the diagnosis of meningococcal disease. Meningococcus (gram negative diplococcus, very distinctive under the microscope) commonly causes meningitis but tends to cause a relatively mild disease with good outcome. It can also cause sepsis that is rapid onset and often fatal – meningitis is rarely a feature of this disease (indeed, having meningitis at the same time is a good prognostic feature).
Recognition
Classic symptoms and signs can be absent, particularly in very young children. Young adults can look surprisingly well. Other conditions can mimic too.
Family/carer opinion esssential where reduced consciousness or communication difficulties.
Babies can have bulging fontanelle, weak, high pitched or continuous crying. Older kids may have aggression or agitation (sometimes blamed on intoxication!).
Check for petechiae in conjunctivae. Tricky in dark skin. Purpura or spreading petechiae are a red flag.
Missed immunisations will increase risk, as will being in group accommodation or recent outbreak.
Diagnosis is by lumbar puncture. Tests should not however cause “clinically significant delay” in starting treatment, and should only be done if safe to do so. Contraindications to LP include –
- extensive or rapidly spreading purpura
- infection at the lumbar puncture site
- risk factors for an evolving space-occupying lesion (see below on imaging)
- any symptoms or signs which might indicate raised intracranial pressure (focal neurology, including posturing/seizures, abnormal pupil responses, GCS=<9 – in which case do imaging first)
Bugs often seen under microscope, which will usually give organism too. Do rapid antigen tests too. White cells will be high (often in thousands if bacterial), protein high (can be over 2 if bacterial). Normal values higher in babies under 3 months. Neutrophil predominance suggests bacterial but this is not v reliable esp in babies. Low glucose v suggestive of bacterial. Presence of blood may indicate alternative diagnosis, or else indicates blood contamination, which should be taken into account [not detailed further, however]
Blood tests should include meningococcal/pneumococcal PCR, HIV test. Do throat swab specifically for meningococcal culture.
Can be complicated by raised intracranial pressure and seizures.
Organisms
In neonates, mostly Group B streptococcus, else gram negative bacilli. Listeria can present with sepsis or meningitis in young infants (90% under 30 days).
In older infants and children, mostly meningococcal disease, else pneumococcal or haemophilus. All declining rapidly as a result of immunisation, currently conjugate Hib, PCV-13 and MenACWY plus 4CMenB.
Treatment
Antibiotics to kill bugs. Steroids to reduce damage.
Out of hospital antibiotics only indicated if likely to be delay in getting to hospital and strong suspicion.
Ceftriaxone is preferred! Broad spectrum, good CSF penetration, once daily. But listeria resistant, and gets chelated by calcium so contraindicated if likely HDU/ICU care where calcium infusions often necessary. Also contraindicated in preterm infants under 41/40 corrected, and in neonates esp jaundice, acidosis, hypoalbuminaemia. In that case use cefotaxime.
If antibiotic allergy, use cef anyway if not severe. If severe, chloramphenicol. Use co-trimoxazole for listeria instead of amoxicillin.
For pneumococcus, 10 days. For Hib, 7-10 days. For GBS, 14 days. For coliforms, 21 days (and discuss using meropenem pending sensitivities). For meningococcus, 5 days only! For unconfirmed bacterial (ie CSF suspicious), NICE says minimum 10/7.
For listeria, amoxicillin or ampicillin for 21 days in total, but discuss using co-trimoxazole, even if not allergic!?). Used to give gentamicin for at least the first 7 days.
Discuss with expert if complicated clinical course.
TB meningitis is a whole different ball game. See NICE NG33 before administering steroids.
Steroids
Dexamethasone has been shown to reduce complications eg deafness in children ≥ 3 months old. Discuss use in babies 28 days to 3 months with infection specialist.
Regimen is 0.15 mg/kg (max 10 mg) qds x 4 days. Ideally given before or with first dose antibiotics – but don’t delay antibiotics. Give if within 12 hours of antibiotics (later than that, only after discussion with specialist).
Stop if bug other than pneumococcus or Hib found. Steroids should not be used in developing countries.
Complications
Hydrocephalus, epilepsy, deafness. Particularly seen with Pneumococcal disease.
Recent evidence highlights that meningitis in early childhood is associated with higher depressive and anxiety symptoms, psychological and behavioural problems, and increased risk of psychotic experiences. Not just that, higher risk of ADHD, and lower IQ on average. Follow up therefore very important for young babies, and probably appropriate to warn families.