In Scotland, now available at Glasgow Private Clinic – see www.peanutimmunotherapy.scot for more details.
More than 10 years ago, shown to be effective in children – 2014 Andrew Clark Lancet STOP II study – 39 children 7-16yrs, peanut flour orally. 2 weekly up dosing, starting with 2 mg, increasing to 800 mg of peanut protein (5 peanuts) then maintained for total of 26 weeks. Tolerance then assessed with challenge.
91% achieved final daily dose (although only 62% actually had negative challenge, which was to 1400mg, nearly double – and previous evidence has suggested that most people at this level can tolerate substantially more with only mild symptoms).
Side effects mainly oral itching (6% of doses), nausea, vomiting. 1 patient needed IM adrenaline (twice) for wheeze. Even minor reactions are annoying especially GI, or if daily! And not always predictable. Tend to get less over time, of course. Paul Turner feels adverse event reporting “massaged” somewhat (no international consensus on reporting) – the word anaphylaxis not used except to thank the Anaphylaxis campaign, yet 22% of patients had wheeze at some point, even though reported rate of wheeze is 0.4% of doses. Still, anaphylaxis in hospital when potential reaction expected not the same as unexpected reaction in community.
Peanut IgE significantly reduced but not SPT! So works as long as you keep taking your daily dose, definitely side effects incl anaphylaxis, but not a cure.
No data yet on rates of reactions after treatment. But using anxiety model, OIT helps with “perception of severity” (as lots of minor reactions), “perceived ability to cope” (as managed already) and “rescue factors” (as more used to carry medicines). Note how experience of using AAI seen as positive (and kids often laugh at anticlimax of it!). 1/3 of benefit of OIT seen after initial confirmation challenge, particularly where anaphylaxis occurred (and in patients as well as parents)! [Sarah Burrell, ADC 2021]
Roughly 10-20% don’t desensitize at all. A further 10-20% don’t achieve full dose. And another 10-20% fail to achieve pass full dose challenge. Benefit likely to be highest for those most at risk of anaphylaxis, yet tendency will be to cherry pick lowest risk cases…
AR101 Oral Immunotherapy trial (PALISADE) – multiple authors including Jonathan Hourihane and George du Toit. US/Europe, N=496 aged 4-17yrs. Dose escalation then 24/52 maintenance (so about 12 months total) with peanut derived product, 67% passed 600mg peanut protein challenge (equivalent to 5-6 peanuts). [Some adult patients included initially but unsuccessful in all of them!]
Frequent mild/moderate events, but also in placebo group! 4.3% had severe events cf 0.8% of placebo group. About 10% withdrew due to adverse events , mostly GI- interestingly another 10% were lost due to parents withdrawing consent, I wonder what their reasons were.
Michael Perkins NEJM editorial – possible risk of eosinophilic oesophagitis? Only 1 case confirmed.
Second trial, just in Europe (ARTEMIS), n=227, up dosing every 2 weeks, just 12 weeks maintenance (300mg), so about 9 months treatment total. 58% tolerated 1000mg peanut (about 8-10 peanuts). Again, lots of mild/moderate adverse events but in the controls too! 1% rate of severe in treatment group (1 patient). 10% withdrew due to adverse effects. No eosinophilic oesophagitis reported. “Clinically important” improvements seen at the time of final food challenge in QOL scores related to “Allergen avoidance and diet restrictions” and “Risk of accidental exposure”, and in FAIM domains relating to perceived likelihood and chance of dying (both self and parent/carer reported) in the future.
Follow up study of self selected group of those who successfully completed 1yr PALISADE programme, who then opted to continue treatment for total of 18 vs 24 months – 48% of former still able to tolerate 2000mg, cf 80.8% of latter. Less adverse events over time of treatment, improved quality of life scores (“clinically meaningful”). Still peanut SPT positive (IgE a bit less for the 24 month group)! There were more accidental peanut exposures in this study than in the original trial, suggesting less vigilance, but the severity of these reactions and need for adrenaline was low, which might confirm an immunomodulatory effect.
Another follow up study (ARC004, Vickery) looked at daily vs non-daily dosing (6 months twice weekly 300mg) after PALISADE. Non-daily dosers had 40% more adverse events, although 80% mild-moderate. Daily dosers had best evidence of desensitization – more than 70% passed 1000mg challenge with NO symptoms (better than PALISADE), and 69% of the cohort (“3A”) who did 2yrs plus could do 2000mg with no symptoms. Evidence of “ongoing immunomodulation” with extended dosing – IgE reduced, SPT did not.
Does not discuss whether non-daily dosing might be appropriate for subgroup who have best immune response to treatment.
ARC0008 study still to report (closed 2023), but provisional data reported show the trend towards decreased adverse events (AEs) at years 1 and 2 is maintained up to 5 years, with 94% of patients experiencing mild or moderate AEs and only 13% discontinuing PTAH use because of AEs overall.
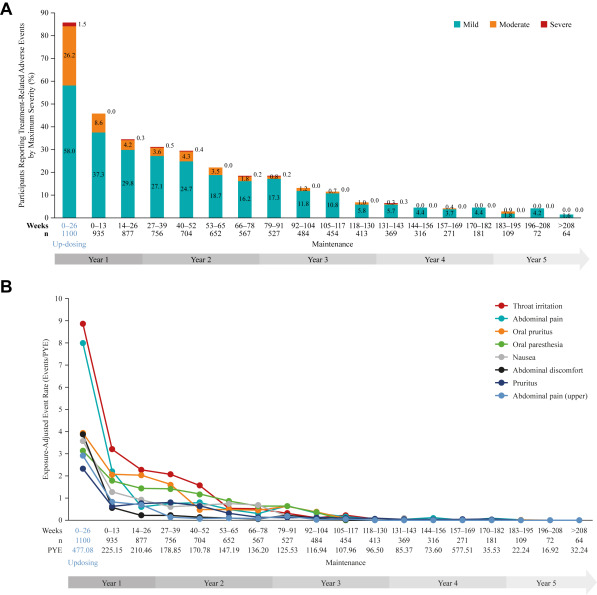
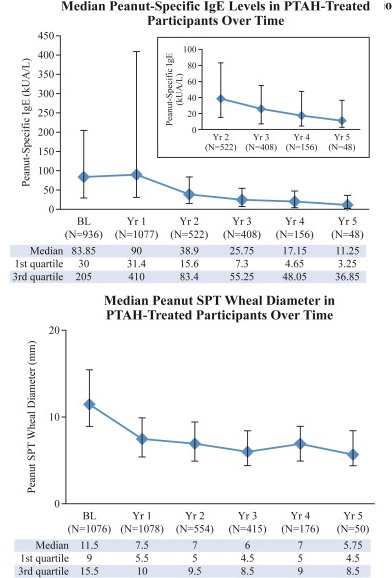
Gastrointestinal symptoms were the most commonly reported treatment-related AEs. A downward trend in systemic allergic reactions was also reported. PTAH treatment resulted in reduced levels of peanut-specific IgE after the first year and increased levels of peanut-specific IgG4, with a lowered peanut-specific IgE:IgG4 ratio. A reduction in median peanut skin prick test wheal diameter was observed (11.50 mm at baseline vs 5.75 mm at year 5). Unpublished draft is available here.
Severe and uncontrolled asthma was an exclusion criterium, obviously. Similarly chronic/recurrent abdominal pain.
AR101 (Palforzia) has now received NICE approval for use in NHS England, which has set targets for patient numbers. In Scotland, SMC did not find sufficient evidence of economic benefit, which fits with ICER report (US economic review) 2019 which showed that peanut immunotherapy led to more systemic reactions and more adrenaline use with no cost benefit. But hard to quantify benefit of reduced anxiety and increased quality of life. Natasha trial currently looking at immunotherapy using commercial peanut flour across several UK sites.
Various forums about immunotherapy. One young person’s blog is Ask About My Peanut Allergy (stopped regular peanut!!! But doing well).
Future may be to combine different routes eg sublingual/patch.